Select a treatment to
get started



























A Smarter Approach to Wellness
Tesamorelin is a growth hormone-releasing peptide potentially showing a significant reduction in visceral fat. It does not replace your hormones. It signals your body to produce more of its own. Targeted. Measured. Effective.

.png)




.png)
.png)
.png)
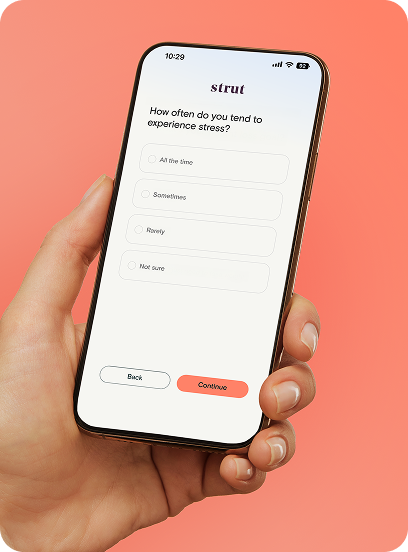

Answer a few questions about your health history, goals, and medications. Takes about 5 minutes.
.png)
A licensed U.S. physician reviews your profile and prescribes Tesamorelin if clinically appropriate.
.png)
Your injection kit ships in discreet packaging. Free shipping. Everything you need included.
Every Strut Health prescription is reviewed and approved by a board-certified physician licensed in your state. Your doctor monitors your progress, adjusts your dosing, and is available for follow-up — included in your plan at no extra cost.
This isn't a supplement you found on Amazon. It's a prescription medication, managed by a real physician, dispensed from a licensed U.S. pharmacy.
.png)
.png)
.png)
.png)



Tesamorelin is a synthetic growth hormone-releasing hormone (GHRH) analog. It signals your pituitary gland to produce and release more growth hormone naturally.

Both stimulate your body's natural growth hormone production, but tesamorelin is more targeted. Tesamorelin may specifically reduce visceral abdominal fat — the deep, metabolically dangerous fat around your organs. Sermorelin has broader GH-stimulating effects but lacks the same level of clinical evidence for fat reduction.

Synthetic HGH directly injects growth hormone into your body, which can suppress your natural production and carry significant side effects. Tesamorelin works through your pituitary gland's natural feedback loop — stimulating production, not replacing it.

Tesamorelin is administered as a daily subcutaneous injection, typically in the abdomen. The needle is small (similar to an insulin syringe). Most people inject before bedtime to align with the body's natural growth hormone release cycle. Rotate injection sites each use.

Clinical studies show measurable visceral fat reduction beginning around 6-8 weeks, with a ~15% reduction at 26 weeks and ~18% at 52 weeks. Individual results vary. Your physician will monitor your progress through lab work (IGF-1 levels) and adjust as needed.

Paroxetine: Nausea, drowsiness, dizziness, headache, dry mouth, sweating, decreased appetite. Most side effects are mild and often improve after 1-2 weeks. Sildenafil/Tadalafil: Headache, indigestion, back pain, muscle aches, flushing, stuffy or runny nose. Apomorphine: Nausea, dizziness, sleepiness.

Tesamorelin is not appropriate for individuals with active malignancy (cancer), a history of pituitary surgery or disease, or women who are pregnant or breastfeeding. Patients with diabetes should be monitored closely, as mild elevations in fasting glucose have been observed. Disclose all medical conditions during your consultation.

Active Ingredients: Tesamorelin

Brand Names: Egrifta, Egrifta WR Drug Class: Growth Hormone-Releasing Factor (GRF) Analog Route: Subcutaneous injection (abdomen) FDA-Approved Indication: Reduction of excess abdominal fat in HIV-infected adults with lipodystrophy
Please read this safety information carefully before starting tesamorelin. This page summarizes key safety data from the FDA-approved prescribing information. It does not include all possible risks, side effects, or drug interactions. Always talk to your Strut Health provider if you have questions about your treatment.
Tesamorelin does not carry an FDA black box warning.
However, the following warnings apply:
Tell your Strut Health provider about all of your medical conditions, including if you:
Age restriction: Tesamorelin is not approved for use in patients under 18 years of age. Safety and effectiveness have not been established in pediatric patients. Tesamorelin is not recommended in children or adolescents because it may cause accelerated linear growth in those with open epiphyses (growth plates).
Tesamorelin may affect how your body processes certain medications. Tell your doctor about all medications you are taking, especially:
General note: Because tesamorelin stimulates growth hormone production, it may modulate CYP450 enzyme activity in the liver. This can affect the clearance of many medications. Always provide your doctor with a complete list of everything you are taking.
Most fluid retention-related side effects (swelling, joint pain, carpal tunnel) are typically transient and may resolve on their own or after discontinuation of treatment.
Immune response: Approximately 50% of patients developed antibodies to tesamorelin during clinical trials. In studies, this did not affect how well the medication worked.

All prescription medications require a valid and complete online consultation prior to approval and final pricing is determined. All compounded medications in the U.S. are considered off-label use. Our licensed doctors have the ability to prescribe compounds for off-label treatment, if they believe that it is an appropriate course of treatment.The drug and medical information provided on this website, is not meant to cover all adverse effects, drug interactions, warnings, medical uses, directions and precautions. The information provided is a medical resource and the judgment of your physician and/or healthcare practitioner should not be substituted. Strut has made every effort to ensure the information on the website is accurate and current based on present medical literature. Strut does not make any guarantees regarding the drug and medical information, and does not assume responsibility regarding the content. The drug safety and drug information contained on the website may be time sensitive and should not be utilized as a medical reference beyond the date hereof. The deficiency of a warning for prescription medications and vitamins provided on this website in no way should be construed to indicate safety, effectiveness, or appropriateness for any given patient. If you have questions or concerns about your medications, please do not hesitate to contact us and/or your medical practitioner, including your pharmacist. Compound prescription products have not been tested or approved by the FDA for their intended use. No claims are made as to the safety, efficacy or use of this compound. Active ingredients used are FDA approved and follow strict United States Pharmacopeia (USP) guidelines when pursuant to a valid prescription.





Medications are prescribed by physicians and compounded by U.S. compounding pharmacies held to strict compounding rules and regulations.
.png)
U.S.
Licensed &
Board
Certified
Physicians
.png)
U.S
Pharmacy &
Medicines
.png)
Discreet & Free Shipping
LegitScript Accredited
.png)
.png)
